-
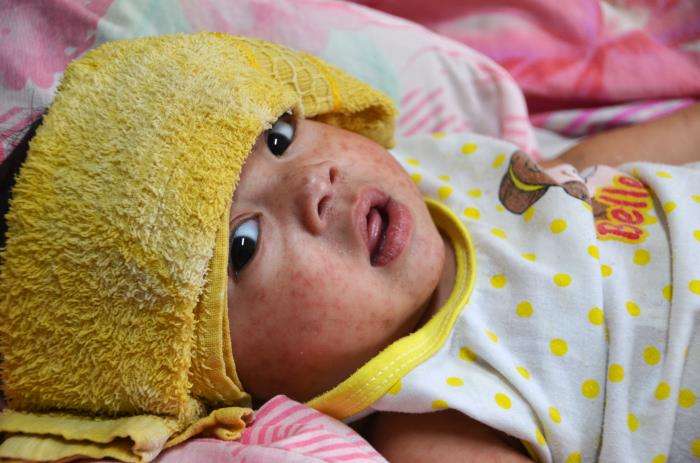
04/20/26 Provider Alert: Measles continues to spread in Washington; Prepare now
Current Situation As of April 15, 2026, a total of 37 cases of measles have been identified in Washington state this year. WA DOH is aware of at least 9 separate introductions of measles into the state since January 1, 2026, all associated with domestic or international travel. Many of these cases have led to…
-
Vaping-associated Pulmonary Illness
CDC Urges Clinicians to Report Cases of Unexplained Vaping-associated Pulmonary Illness Multiple cases of severe pulmonary disease in patients who use vape products have occurred in Wisconsin and Illinois. Requested actions Ask patients who present with respiratory or pulmonary illness if they vape THC or e-juice. Determine what kind of THC or e-juice they’ve vaped,…
-
-
Hepatitis A Fact Sheet
Overview Transmission: Fecal-oral. Incubation Period: Average 28-30 days (range 15-50 days). Symptoms: Anorexia, vomiting, abdominal pain, fatigue, followed by jaundice. Severity of illness increases with age. Infectious Period: 14 days prior to onset of jaundice to seven days after onset of jaundice. Infants can excrete virus in the stool for longer periods of time. Epidemiology:…
-
“What is Public Health?” from American Public Health Association
Learn why public health is essential in American Public Health Association’s “What Is Public Health?” article. Read Fact Sheet →
-
Hand Hygiene In Healthcare Settings
What is hand hygiene? Hand hygiene refers to the use of hand washing with soap and water or alcohol hand sanitizer (60% alcohol or greater) in order to reduce infection rates, reduce transmission of antimicrobial resistant organisms and stop outbreaks of communicable disease. Why is hand hygiene important? When should I use hand hygiene? How…